
Water in Nature and Possible Industrial Uses
By
Roberto Triolo and Irene Ruffo
Dipartimento Di Chimica Fisica – Università di Palermo –
Palermo (Italy)
Liquid
water (H2O) is the most common substance in the world. Despite being
apparently so simple, and therefore despite being considered an ordinary
material, water is able to display a variety of different properties which make
it the most important material in Nature. If we think how our day starts and
how ends, how our days progress, we shall probably realize that most of our
activities depend on water. The water
molecule is very simple and small. Only three atoms make it: two Hydrogen atoms
(H) and one Oxygen atom (O). However H and O are quite different atoms, the
first one being very electropositive (has tendency to acquire a positive
charge) and the other has tendency to acquire a negative charge. However, the
extraordinary complexity of its properties is also due to its size; on the
other hand these properties seem to fit ideally into the requirements for life
as no other molecule. All the most interesting properties seems to derive from
the possibility of the opposite charges to interact generating a peculiar type of
“bond”: the Hydrogen Bond (H-bond) which is responsible for the high specific
heat, the unusually high heat of fusion and heat of evaporation, the high
melting and boiling point, and the possibility of acting as solvent for polar
molecules in its Normal Water conditions (NW) and as a more “organic type”
solvent in Near Critical (NCW) and Super Critical (SCW) conditions. H-bonds are formed as the Hydrogen atom
(the positive end of the molecule) is attracted from the Oxygen atom (the
negative end of the closest water molecule). In these conditions a network of
water molecules is formed. The net result is similar to having a macromolecule
made by many single molecules held together. Water molecules are pretty much
held together by these forces, so water surface is very resistant to
penetration. Chemists call this property “Surface Tension”. In fact, any
object, to penetrate the surface, must break this network of H-Bonds. A
difficult task! It is well known that ice is lighter than liquid water: also
this property is a consequence of the H-bonding, which create voids in the ice
structure, and therefore a given volume of ice will weight less than the same
volume of liquid water, which is heaviest at 4°C. Below and above this
temperature water is lighter. The consequence is that lakes and oceans freeze only
on the surface and not from the bottom. Without this property life would not be
possible, neither the cycle of water would be possible. Water has a large heat
of evaporation because, to evaporate, H-Bonds holding molecules together must
be broken. This can be done, but only if large amounts of energy are used and
so water has the largest heat of evaporation of any other material liquid at
room temperature in Nature; water becomes solid at 0°C and boils at 100°C and
therefore is liquid in the vast majority of the earth surface. When compared
with the gas phase (water vapour vs. air), one may notice that water warms up
more slowly and cools down more slowly than air. This makes life more
comfortable for the living creatures, as our body temperature is basically
constant, despite the seasonal and daily changes of the atmosphere. In the
table shown in the next page some of the properties of water are reported
together with their relevance to environmental issues. Finally the strong
H-bonding which characterizes the solid form of water makes ice a very peculiar material. It is
possible to slide on it, to skate on it, to make it act as heat sink, helping
to maintain an average temperature ideal for life processes. Let’s now assume
that we are breaking these bonds. When the
H-bonds progressively break, water loses more and more the characteristics of
solvent able to solubilize salts, acids and many of
the basic chemicals necessary for life (“polar solvent”), and starts to become
more like typical organic solvents (“apolar”
solvents). So, while at room temperature and pressure water is a poor solvent
for plastics, in Supercritical conditions (high temperature and high pressure)
becomes a good solvent for plastics. In turn this means that water (which is
not dangerous for the environment) may replace solvents like benzene and Fluoro-Chloro-Hydrocarbons which are used in many
industrial processes and are very dangerous for the environment. In a sense we
may consider water as a sort of universal solvent, whose properties can be
changed at will by changing its physical conditions (T and P), so one may expect dramatic changes in the solvating
properties of water, moving across the phase diagram. In view of these
properties, it is possible to find applications for supercritical water (SCW) in eco-friendly processes. For example many organic molecules, as well as O2, N2, CO2,
are miscible in all proportions with water under these conditions. This makes SCW a good medium for combustion reactions
, while it may dissolve organic materials, like waste materials, garbage
etc. This opens a variety of possible processes.
Reactions and Phase Equilibria in Near-critical Water (NCW)
In the temperature
range of 250 to 350 °C (near-critical) water is an environmentally-benign
solvent for a wide variety of manufacturing processes in the chemical,
petrochemical, pharmaceutical, and plastics industries.
|
Property |
Comparison with other molecular fluids |
Impact on the Environment |
|
Specific Heat = 4.18 J/g.K |
The highest, except NH3 |
Makes T changes gradual Makes heat transfer, through water, possibile
and very extensive Helps keeping constant the body T. |
|
Heat
of Fusion = 333 J/g |
The highest, except NH3 |
Help transfer of Heat through melting and solidification (Cycle of Water). |
|
Heat
of vaporization= 2250 J/g |
The highest known |
Help transfer of Heat through vaporization and condensation (Cycle of Water). |
|
Surface Tension =
7.2 X 109 N/m |
The highest known for liquids |
Important
in Cell Physiology. Formation
and shape of liquid drops |
|
Viscosity
= 10 -3 N.s/m2 |
Smaller than the majority of liquids at comparable temperatures |
Easy flow properties |
|
Dielectric
Constant = 80 at
20° C |
The highest among liquids (except HCN and
H2O2) |
Keeps
ions apart in solution |
In general NCW has the
following advantages:
· Replaces many less desirable
solvents, such as aromatic hydrocarbons and chlorinated compounds, reducing the
environmental impact.
· Avoids the use of polluting mineral
acids and hazardous catalysts.
· Permits the minimization or even the
elimination of unwanted by-products.
· Provides better control of reactions
which can be run homogeneously instead of heterogeneously.
· Facilitates closed processes by reuse of
waste material.
· Provides simple separation after
reaction.
Reactions and Phase Equilibria in Super-critical Water (SCW)
The chemical and
physical properties of SCW, makes it an ideal medium
for oxidation reaction of organic materials and their mixtures wit/without eteroatoms (halogens, sulphur, phosphorus, etc). The final products
would be water and carbon dioxide for the organic portion, acids, bases and
salts for the remaining portion. All these products can be easily separated and
therefore pollution is very low. For this reason over the past decade, a major
research effort has been focused on the destruction of toxic organics by means
of total oxidation in SCW. The process is highly
effective but there can be serious problems of corrosion associated
with large scale waste destruction, so serious indeed that many chemists
have been discouraged from even contemplating possible uses of SCW as a medium for chemical reactions. Recently, however,
there have been a number of reports which show that high temperature and
supercritical water can be used constructively for reaction chemistry.
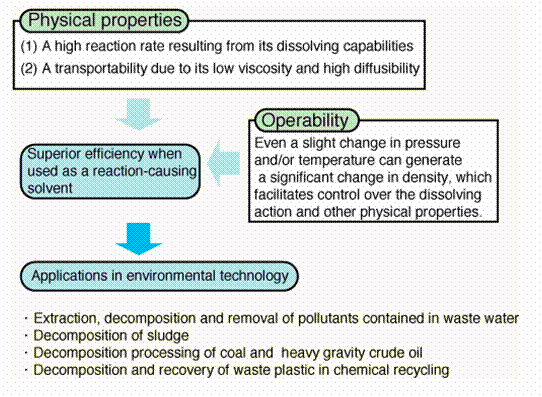
The figure summarizes the main advantages of using SCW
as reacting medium. Also a list of possible applications which begin to be
explored is presented.
We wish to end
this presentation reminding that Nature has already applied most of the “techniques”
and “approaches” presented in this paper. Most geological fluids, in fact,
contain water and carbon dioxide in supercritical conditions. This permits all
the chemical transformations which give life to a variety of geochemical
materials. It is for this reason that scientist have
begun very recently to investigate not only NCW and SCW, but also mixtures of supercritical fluids, with water
and CO2 as primary components.